THE MOLE CONCEPT. Objects are usually measured by counting or by weight (mass) It's easier to measure large objects by counting –Elephants, cars, eggs. - ppt download
Chemistry: Atom Molecules Arithmetic: Importance and Scope of Chemistry- (For CBSE, ICSE, IAS, NET, NRA 2024) FlexiPrep

Analysis of Two Definitions of the Mole That Are in Simultaneous Use, and Their Surprising Consequences | Journal of Chemical Education

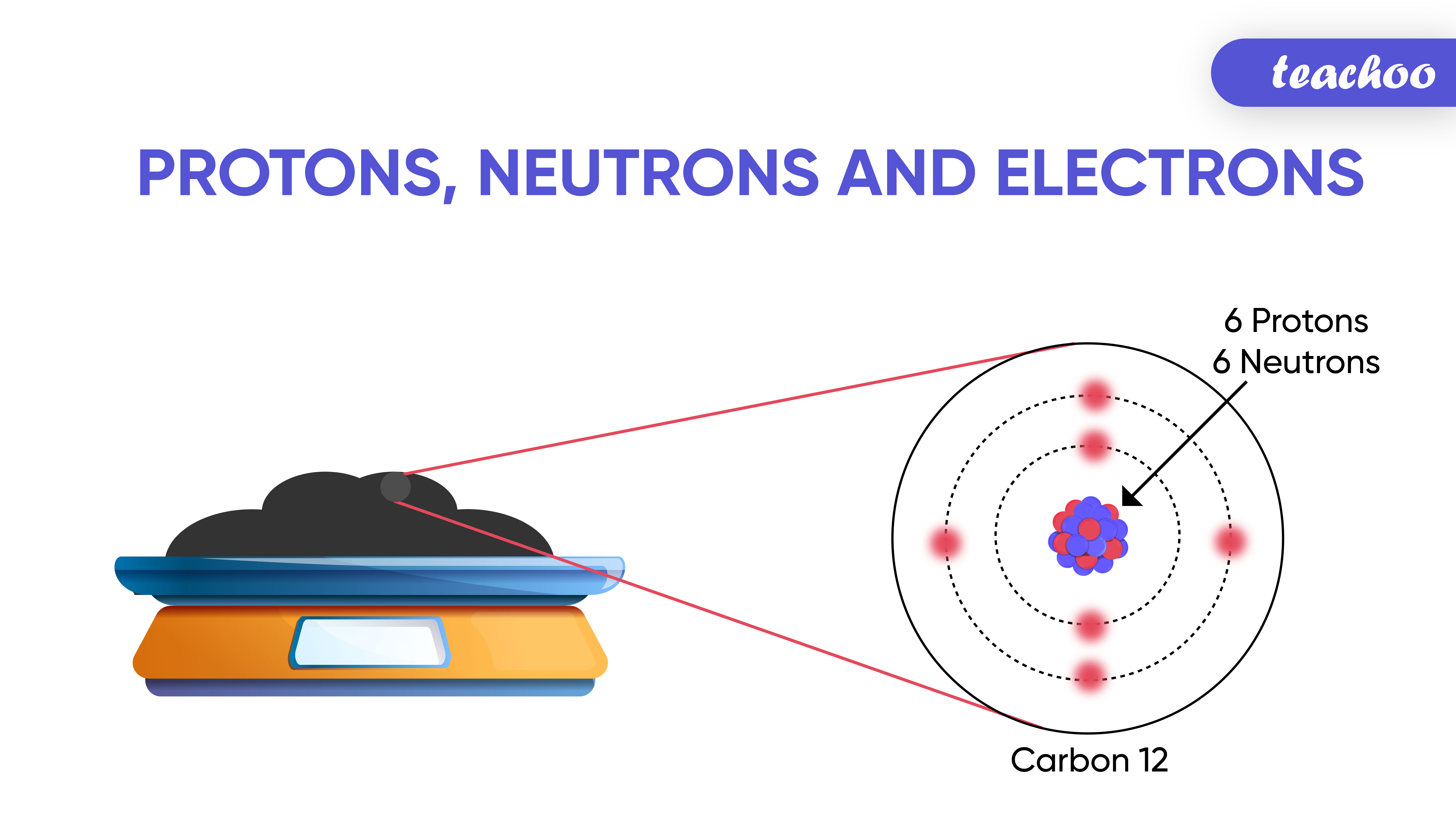
Moles. Definition A mole is the mass of a substance which contains the same number of particles as 12 grams of the isotope carbon 12. These particles. - ppt download


















:max_bytes(150000):strip_icc()/what-is-a-mole-and-why-are-moles-used-602108-FINAL-CS-01-5b7583f6c9e77c00251d4d68.png)