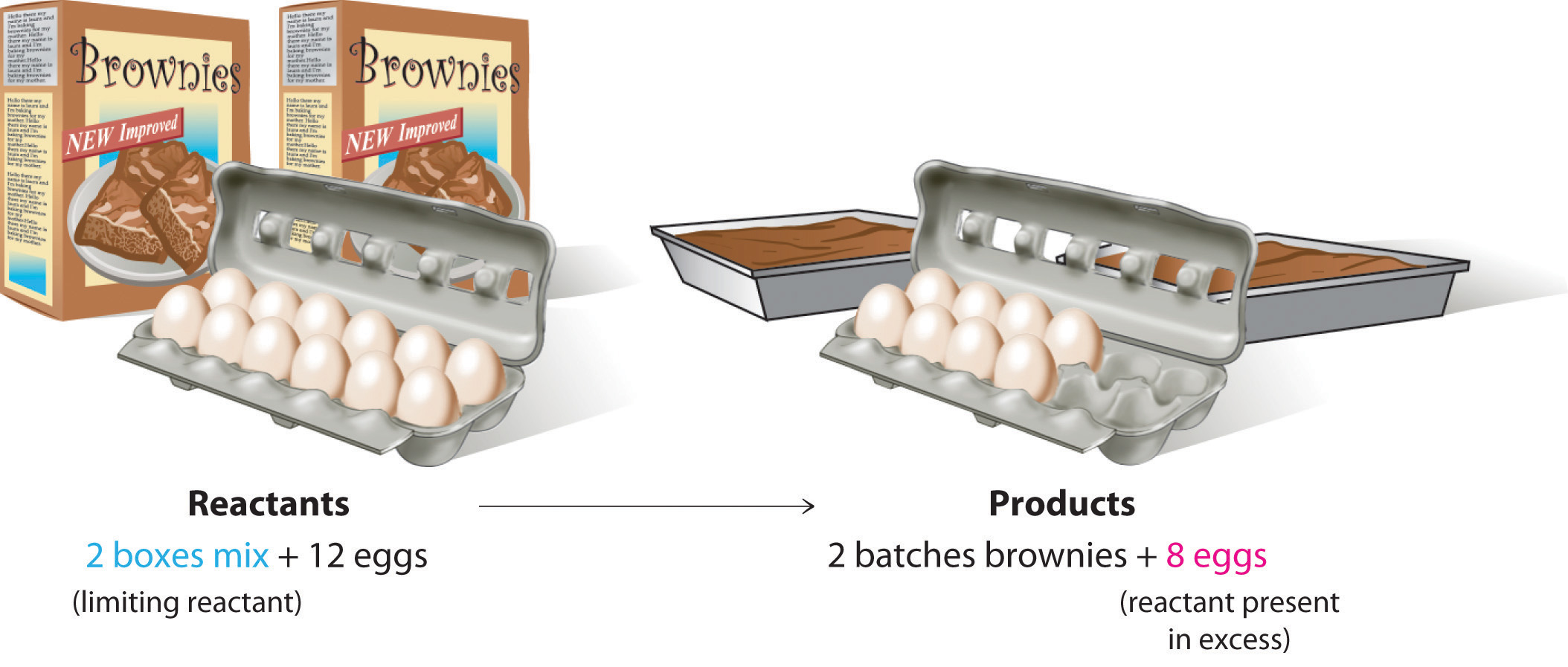
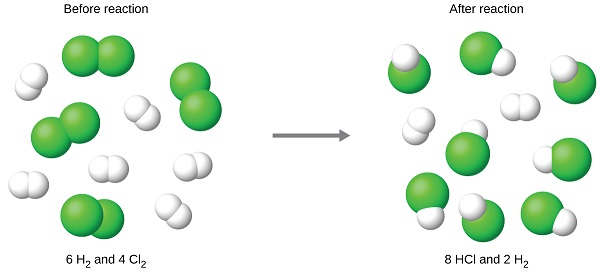
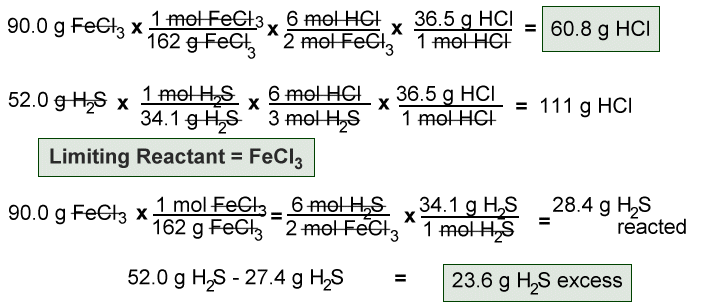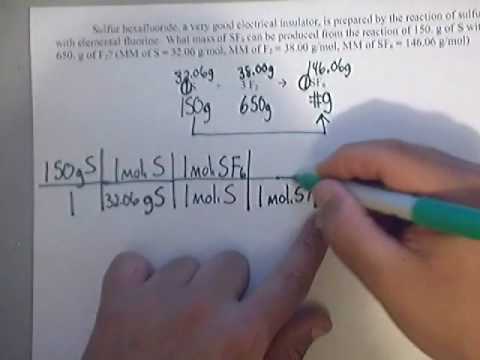Limiting Reagents Determine the mass of P 4 O 10 formed if 25g P 4 and 50g O 2 are combined. P 4 + 5O 2 P 4 O 10 What

SOLVED: Calculate the Limiting Reactant and Remaining Substances 3. Given the amount of each reactant in the table below, determine the limiting reactant, the grams of copper metal created and the amount

How to Find How Much Excess Reactant Remains Examples, Practice Problems, Questions, Summary - YouTube
![Grade 11: Stoichiometry] Is there not a need to find out the limiting reagent when we calculate the mass of ammonia? : r/HomeworkHelp Grade 11: Stoichiometry] Is there not a need to find out the limiting reagent when we calculate the mass of ammonia? : r/HomeworkHelp](https://i.redd.it/4abku36f34j51.jpg)
Grade 11: Stoichiometry] Is there not a need to find out the limiting reagent when we calculate the mass of ammonia? : r/HomeworkHelp