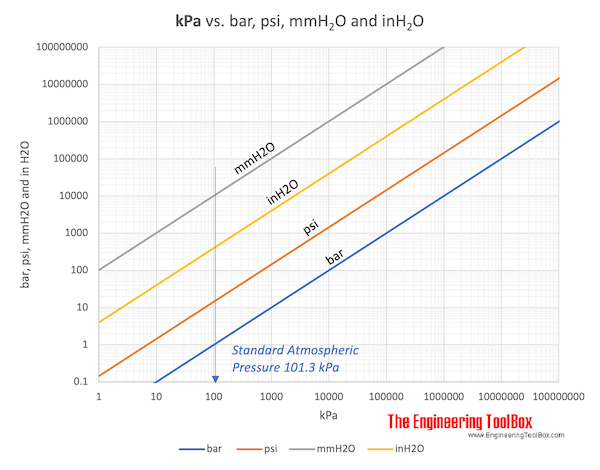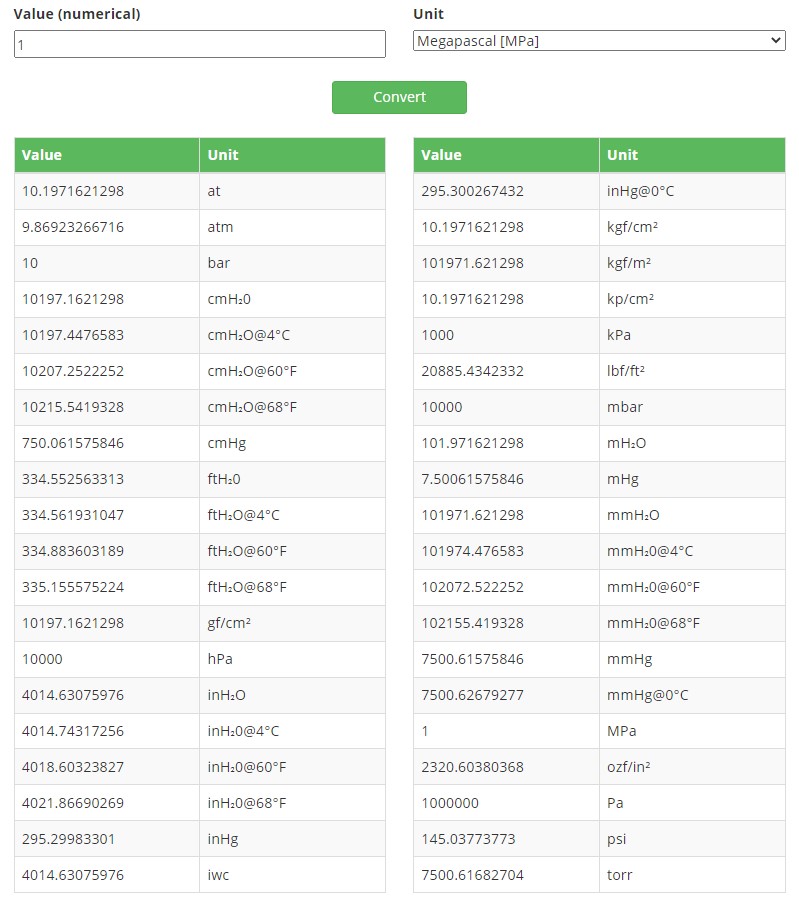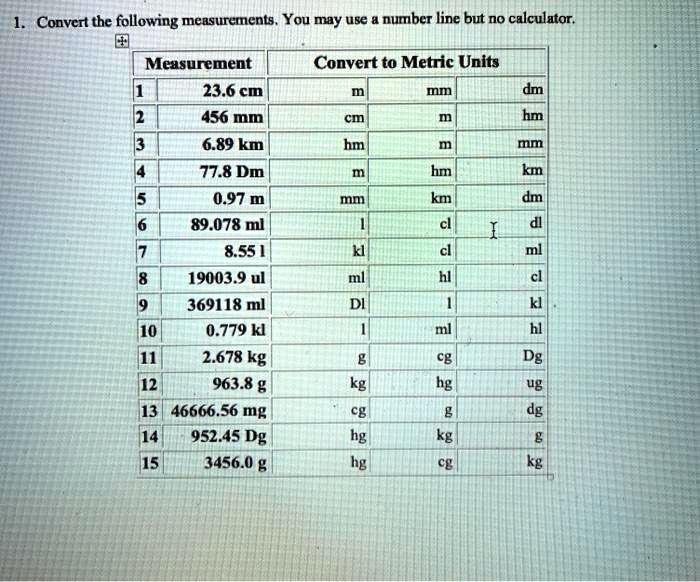
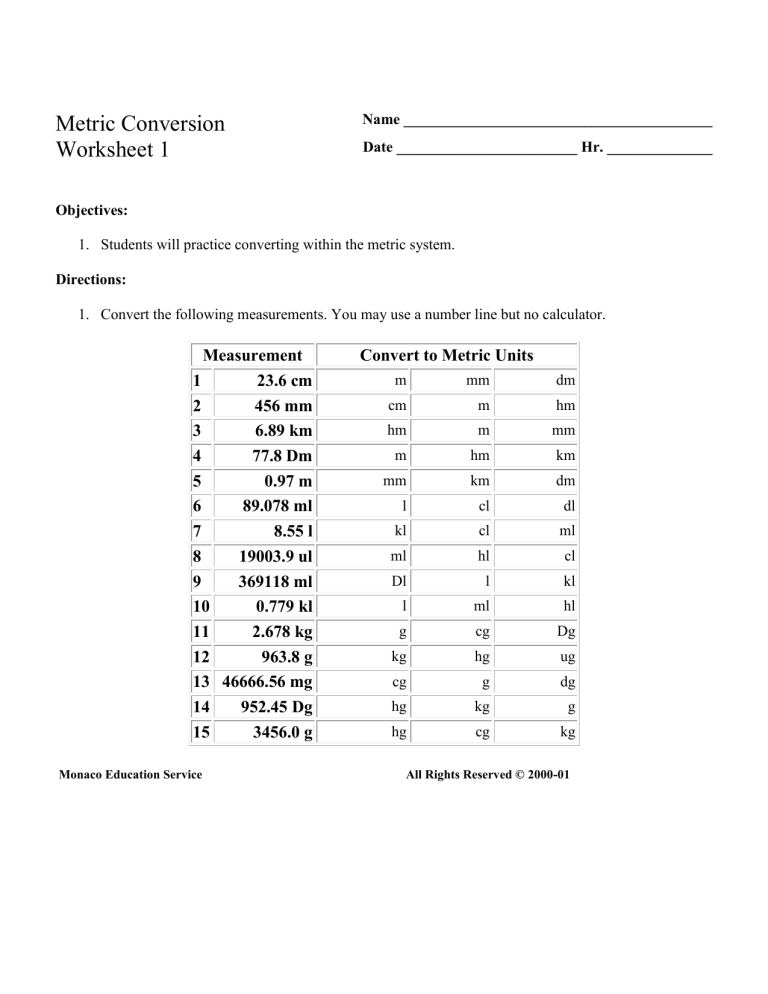
SOLVED: Convert the following mensurements . You may use number line but no calculator Measurement 23.6 cm 456 mm 6.89 km 77.8 Dm 0.97 m 89.078 me 8.55 [ 19003.9 ul 369118

Converting Length Units Length Units Conversion Stock Vector (Royalty Free) 2060867282 | Shutterstock

Calculate the ppm of mercury in water in given sample contain `30mg` of `Hg` in `500ml` of solution - YouTube

At 298 K, the vapour pressure of water is 23.75 mm Hg. Calculate the vapour pressure at the same temperature over 5% aqueous solution of urea (NH2CONH2).

The vapour of a sodium containing 13 × 10^-3 kg of solute in 0.1 kg of water at 298 K is 27.371 mm Hg . Calculate the molar mass of the solute.

La Wiphala de Osler 🇵🇪 on Twitter: "@MikeMD2486 Maestro, a eso me referia sino q no lo he dicho asi, con q la ecocardio es escencial, sobretodo para hacer el score de

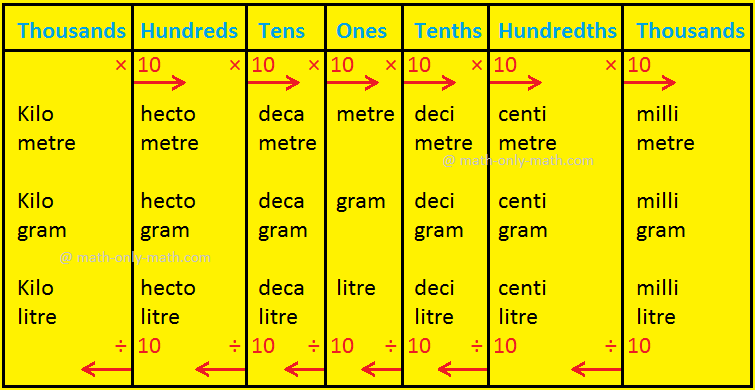
Metric Prefixes and Unit Conversions | Useful Equations And Conversion Factors | Electronics Textbook




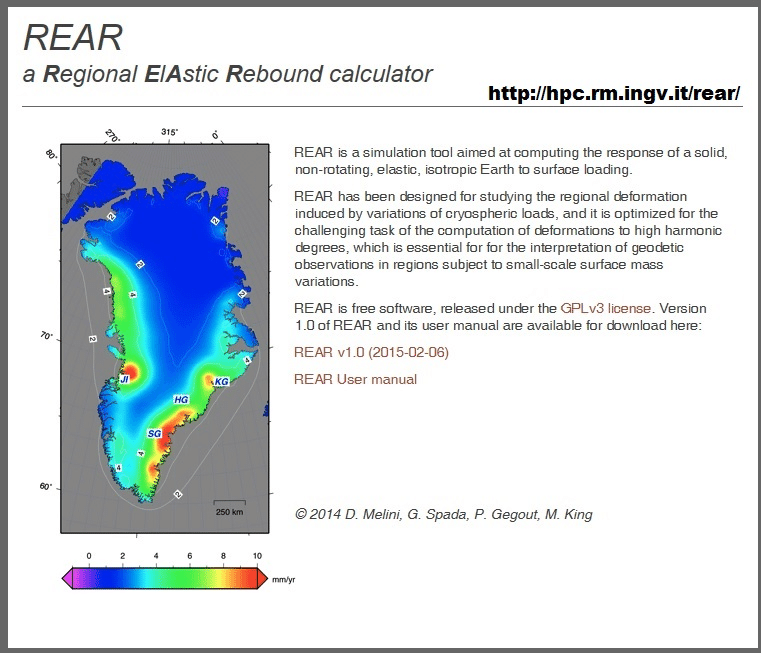
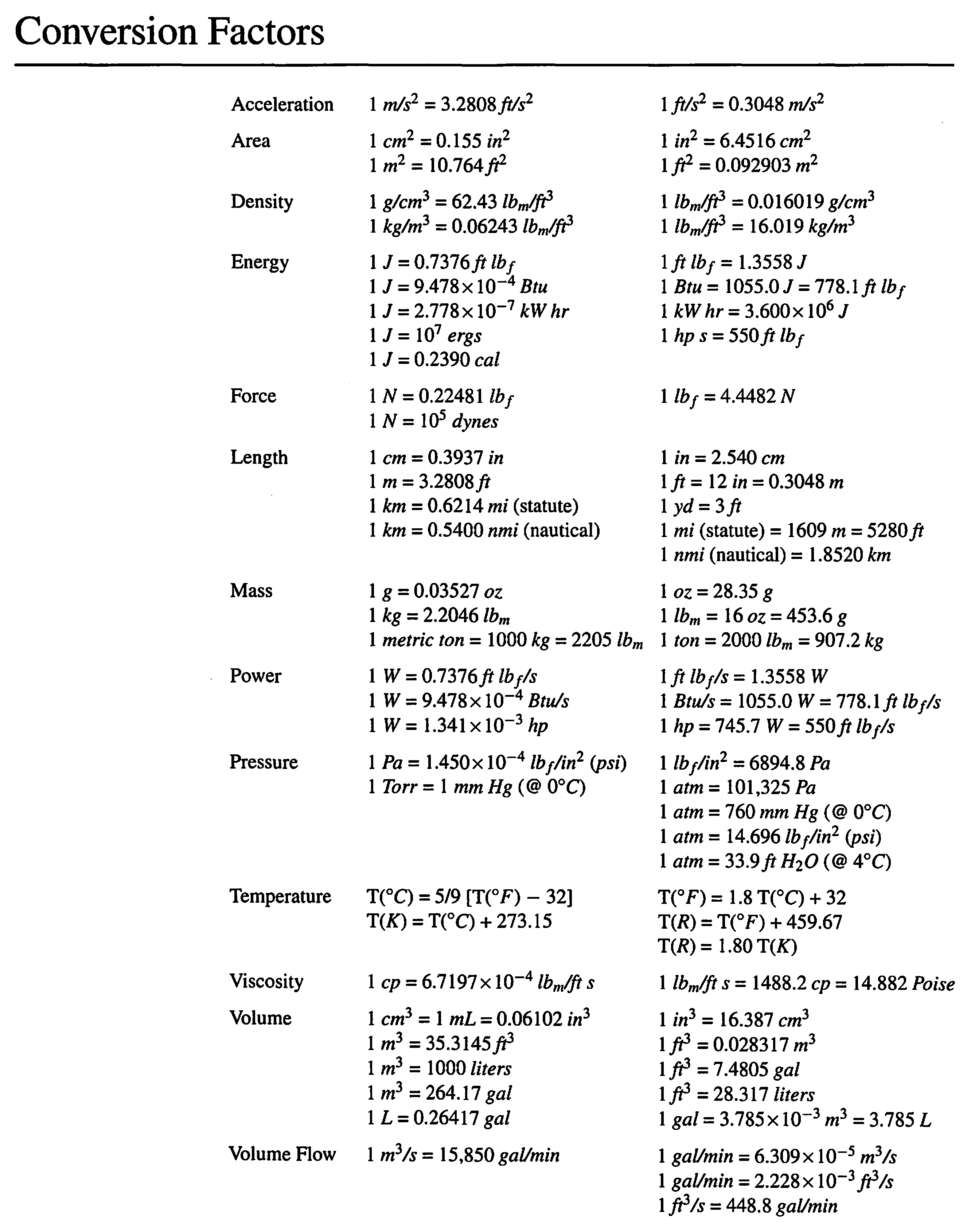

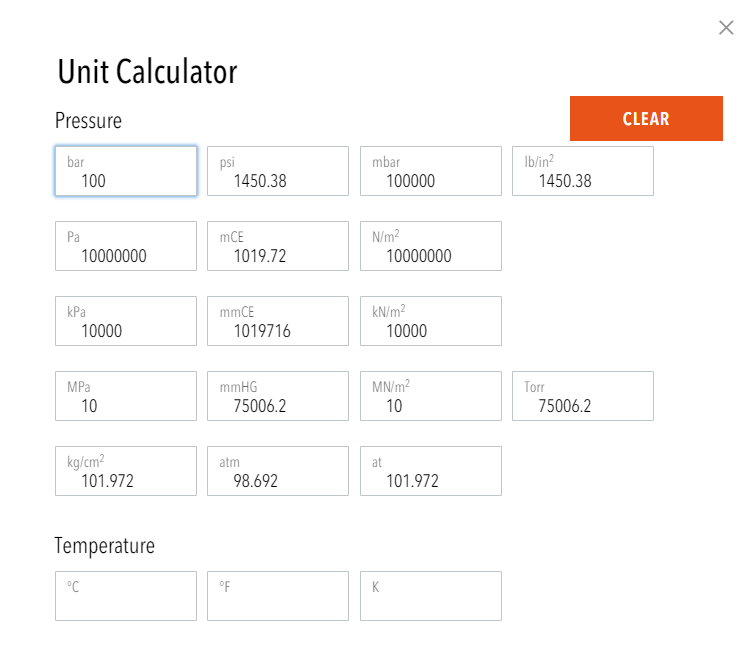

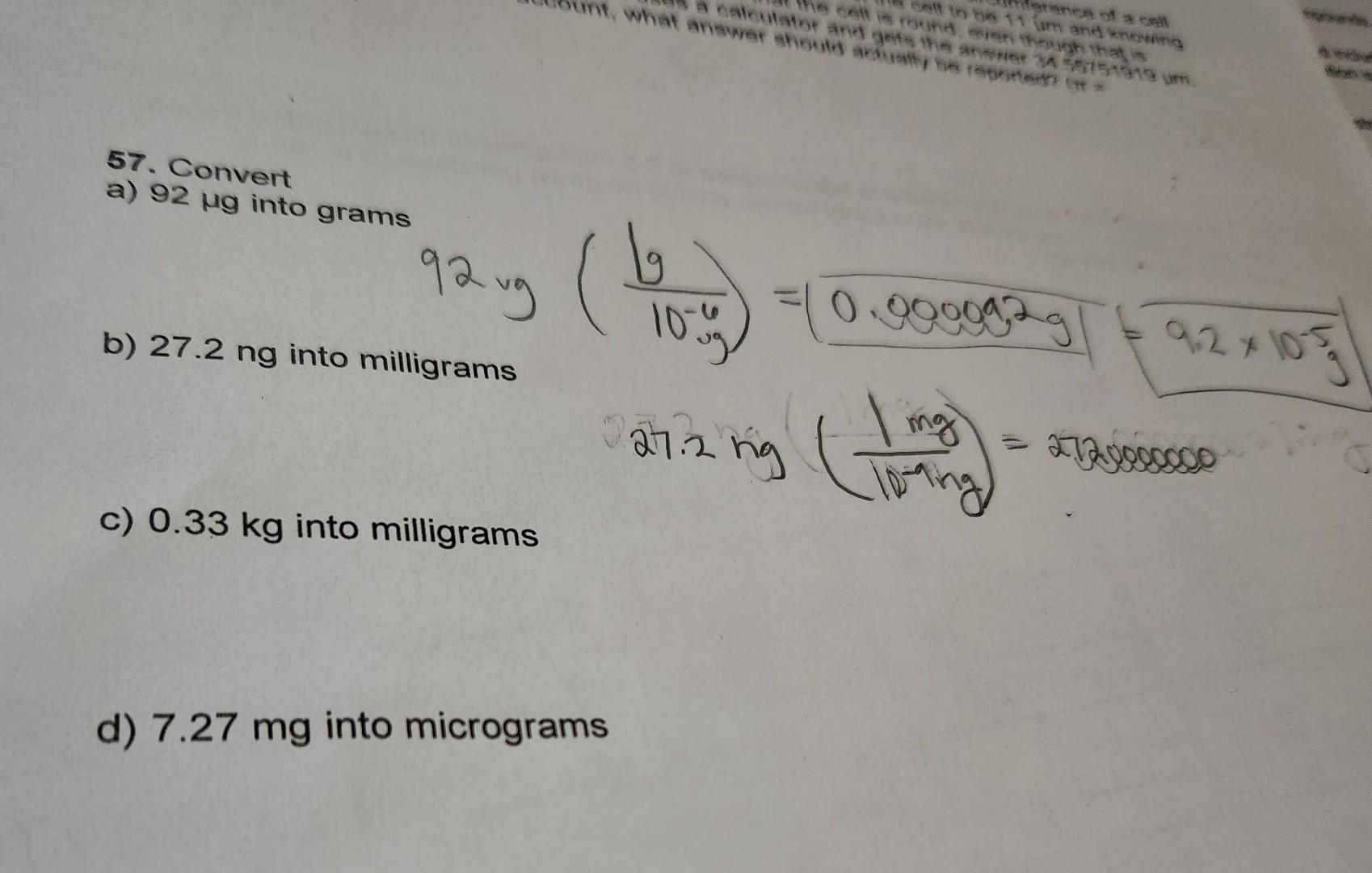

![Kilograms [kg] - YoosFuhl.com Kilograms [kg] - YoosFuhl.com](https://www.yoosfuhl.com/wp-content/uploads/2019/06/kilogram-charts.jpg)